|
We have been working diligently in Pre-AP to learn about how to write electron configurations and orbital notations. I am not allowed to post the answers to what we have been working on lately, but I assure you that the students have caught on well! There will be a quiz on these concepts early next week. Don't forget to do your homework!
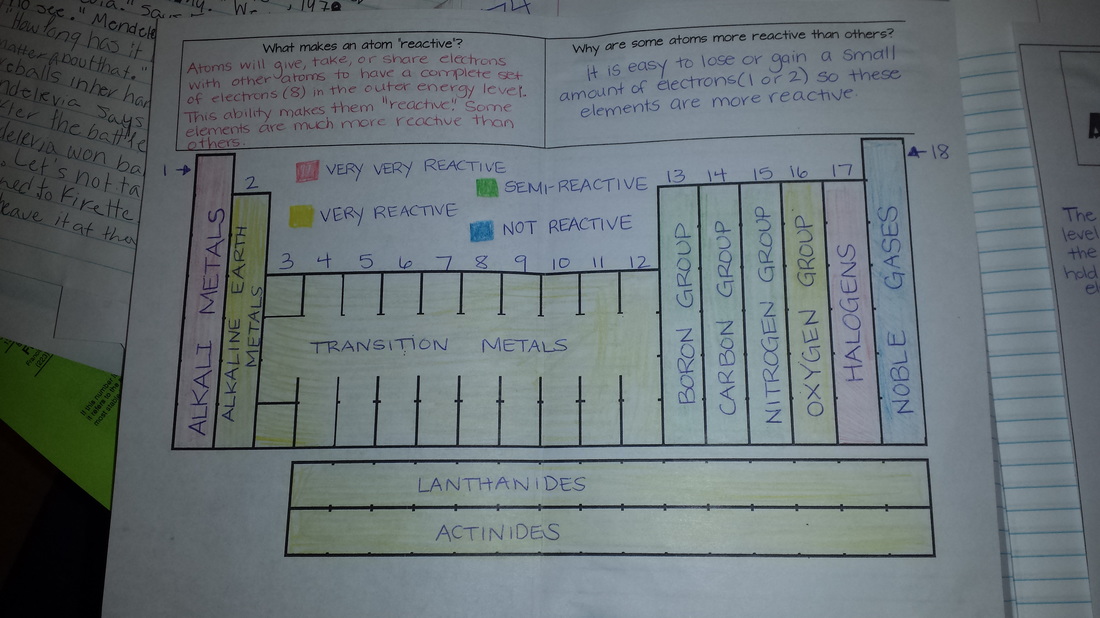
I did not feel like the Outlines were doing the information justice, so we are currently going back to delve into atoms a little bit deeper. Friday we worked on the reactivity of the periodic table and valence electrons. Today we started electron configurations. I will post electron configurations AFTER we have finished the lab. There will be a quiz on Electron Configurations on Wednesday!
Don't forget to study your polyatomic ion flashcards! Today we worked on finishing notes for changes of state. We then discussed freezing and melting before getting back into the discussion about bubbles. Bubbles contain both the liquid and gas phases. You have to blow (gas) through a liquid layer to create a bubble. This also led us to a discussion about surface tension and how beginning with a rectangular frame will still yield a round bubble. Check out the notes below if you missed them as well as some bubble recipes to try over fall break! We will be doing Data Set Question 6 on Wednesday when we get back. The outline is posted below!
It appears that the notes I had set to post yesterday have magically disappeared! Therefore, I am reposting those notes in addition to what we did today. Today was a BLAST and we explored phase changes, specifically sublimation with dry ice. Yesterday students used a card sort to learn about solids, liquids, and gases and were asked the question "Are bubbles a solid, liquid, or gas?" This, of course, brought on a huge debate! More on that later! Check out the notes and pictures below! Today we started research for the periodic table project. Project details are listed below. Most of the research and development for this project will be completed in class, but students who do not use their time wisely or miss class will need to complete it at home. This project is due on October 16th! I will give 5 BONUS points to anyone who turns it in on Thursday, October 15th! 1. Identify the number of protons in Phosphorus.
The atomic number of Phosphorus is 15 so that tells me there are 15 protons in the nucleus. 2. How many electrons are in Potassium? Potassium's atomic number is 19 which is equal to the number of electrons. 3. How many neutrons are in an Argon atom? Argon's mass is 40 (39.95) and it's atomic number is 18 so when I subtract, I get 22 neutrons. 4. What is the atomic mass of Aluminum? According to the periodic table, the mass is 26.98. 5. What is the atomic number of Sodium? According to the periodic table, the atomic number is 11. 6. What is the chemical symbol of Mercury? Hg 7. What is the name for Xe? Xenon 8. Which element has the greatest mass, Fluorine or Sulfur? Fluorine's mass is 19 while Sulfur's is 32.07 so Sulfur has the greatest mass. 9. Which element has the most protons, Silicon or Beryllium? Silicon's atomic number is 14 and Beryllium's is 4 so Silicon has the most protons. 10. Which element has the most neutrons, Helium or Carbon? Using Mass-atomic number I found that Helium has 2 neutrons while Carbon has 6. **Also make sure you study the location of the metalloids, metals, and nonmetals! ** |
What Is
|
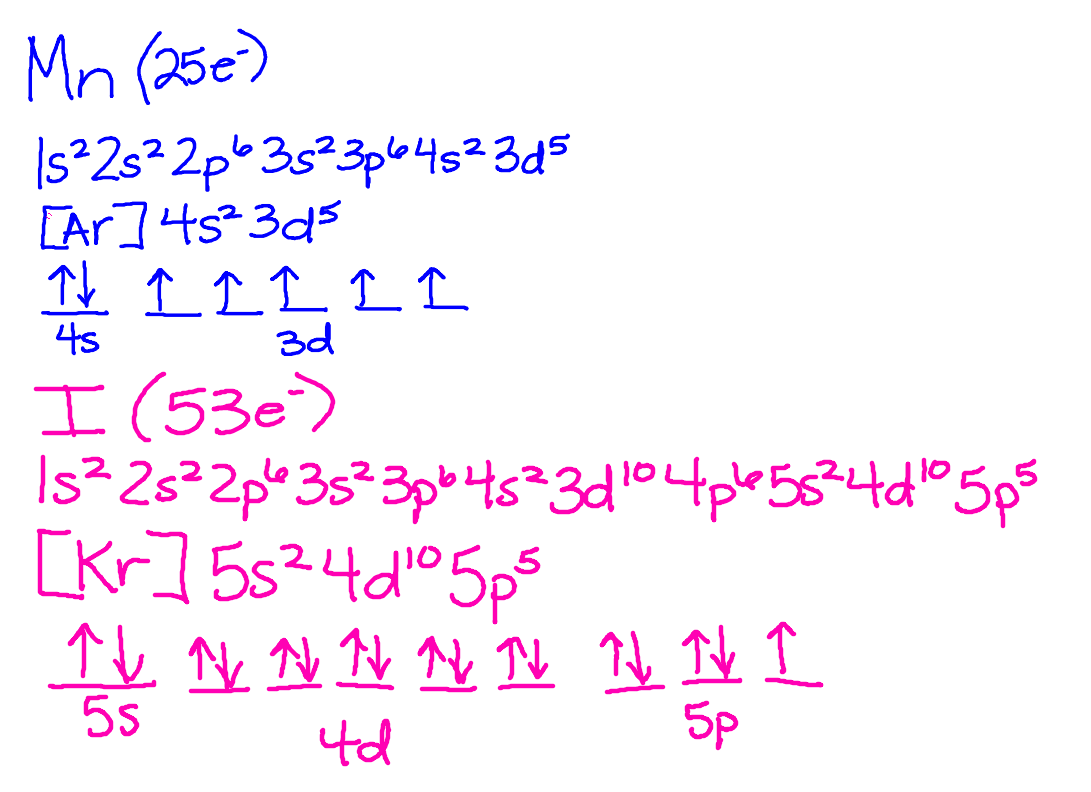











 RSS Feed
RSS Feed