|
Today we talked about moving literal equations, specifically the equation for density. I am adding an instructional video below that explains how to move literal equations if you still have questions. We will continue to work on this concept throughout the year, and students will also see this concept in math. We will continue working with density on Tuesday before we start reviewing for the test on Wednesday.
0 Comments
I apologize for the lack of posting yesterday, but I completely forgot! Yesterday we worked with dry ice and observed what happens under specific conditions. Mr. Byrd even stopped by to play for a while! Today we reviewed chemical and physical properties and changes. We also started density. I am posting the notes for density below!
Today students did a little informational reading to help them determine the differences between physical and chemical properties and changes. They completed a worksheet in class with their group using an outline and the textbook, as well as their ideas as a group. They were then given a creative writing assignment to begin. This creative writing assignment is due on WEDNESDAY at the beginning of class and is posted below for students who missed it!
Today students received two outlines. The first covers states of matter (solid, liquid, gas) and the second covers physical and chemical properties and changes. There will be a data set question on 2.5 on Monday of next week. We will have our Unit 2 test on Thursday, October 6th. It will cover all of the Unit 2 outlines as well as the experiments and activities in class. I am attaching the outlines below if you need them!
Today students took a pop quiz on atomic structure and finding the number of protons, neutrons, and electrons using the periodic table. Ms. Hendricks will be in here tomorrow going over Cornell notes and study habits, so we will continue with Bohr Models on Friday!
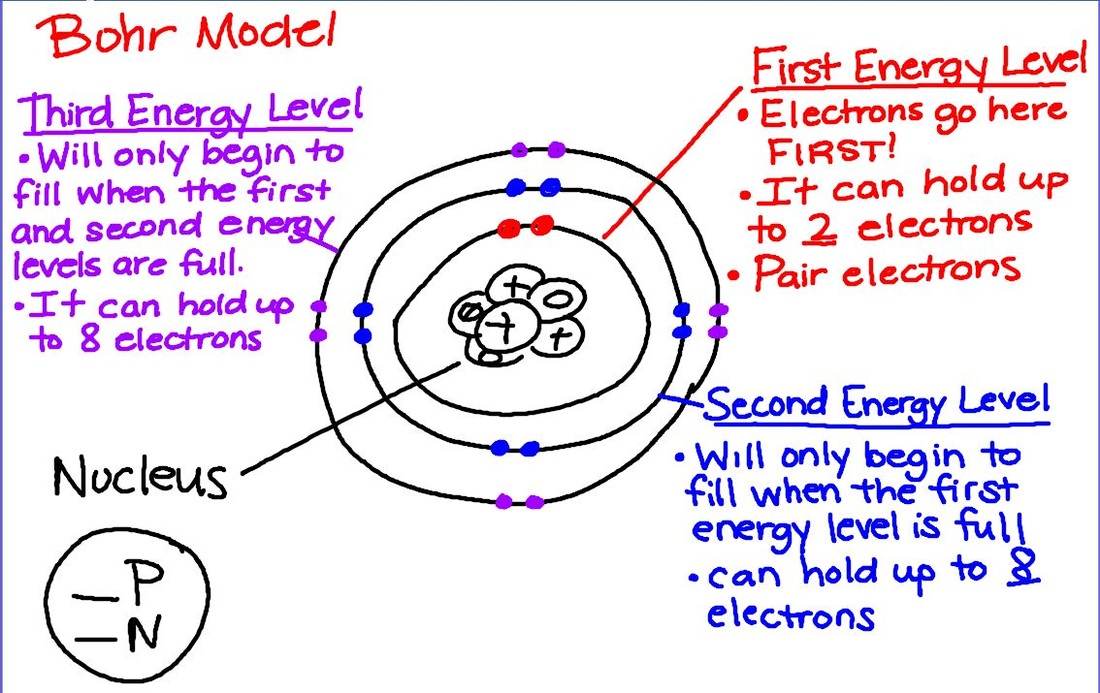
Today we began looking at atomic structure by creating Bohr models of the first 18 atoms. We modeled the structure using beans today and we will draw them beginning tomorrow. We will also begin looking at electron configurations for specific elements using the Aufbau principle.
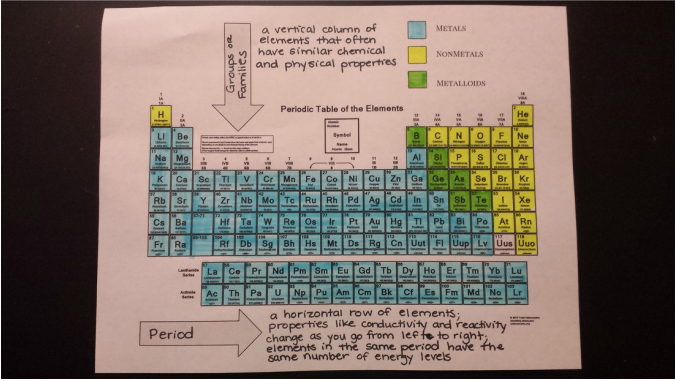
Today we color coded the periodic table to represent the metals, nonmetals, and metalloids and their locations. Students also received homework on finding the number of protons, neutrons, and electrons for neutral atoms that will be checked tomorrow. We will continue using the periodic table to draw Bohr models tomorrow!
Good afternoon! Today we took a pop quiz on the Atomic Theory timeline and completed one data set question from the notes. Many students are now concerned about their grade in Pre-AP. Pre-AP requires a GREAT DEAL of responsibility on their part. The Atomic Theory outlines were given out on Thursday of last week and I did my best to ensure that everyone had it. For the pop quiz today, many students complained that they had not studied so I gave some time in class. As a Pre-AP student, it is essential that you read the outlines and study them to prepare yourself for what we do in class. Even if I do not directly go over it, you are still expected to know it if it is on an outline. The point of Pre-AP is to APPLY the knowledge gained from reading the outlines at home in class. This is virtually impossible to do if you are not completely familiar with the information in the outlines. Most students find they have to read them more than once and even write their own notes from the outlines to be successful. YOU are in charge of YOUR learning. It is your decision to make the most of what you have. You only have to do it!
https://phet.colorado.edu/sims/html/rutherford-scattering/latest/rutherford-scattering_en.html
Above you can find a great interactive we used in class that allows students to compare Rutherford and Thomson's work and help see how Rutherford got the results he did during his gold foil experiment. We are now moving on to the periodic table of elements! Today in science we work on identifying shapes under a piece of cardboard using only a marble. Similar to Rutherford's work, students had to deduce the shape based on how the marble bounced off of it. Students made observations and educated guesses about the shape before we revealed them at the end. We will continue with a simulation on Rutherford's experiment tomorrow! |
What Is
|
||||||||||||||||||||||||


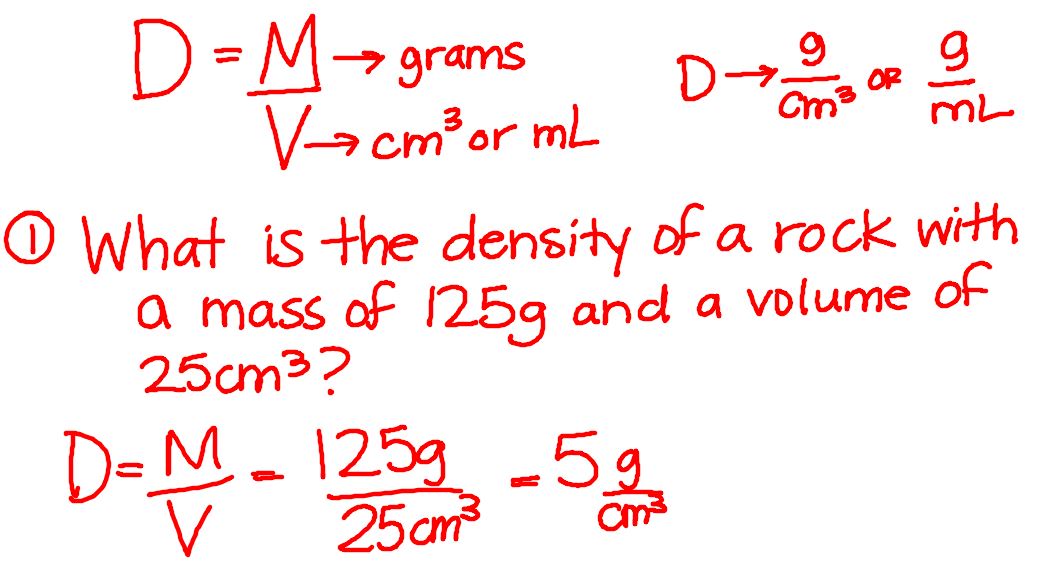


 RSS Feed
RSS Feed