|
Today we started class with this great video about how osmosis changes the sizes of gummy bears. Students then had to hypothesize how two different liquids that were NOT water would change their gummy bears. In each class we are testing Coke, Diet Coke, Sprite, Diet Sprite, Gatorade, G2, Powerade, Powerade Zero and Sparkling water. Students examined the ingredients and nutritional info for each liquid before recording a hypothesis. Now we just wait to see what happens! Check back tomorrow for the results of the experiment!
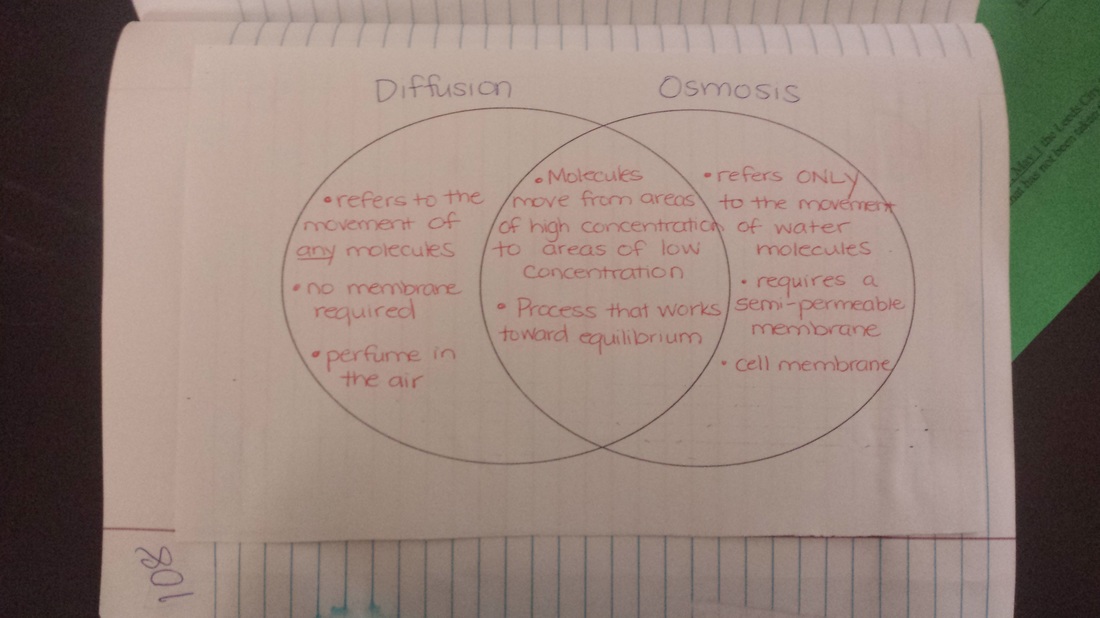
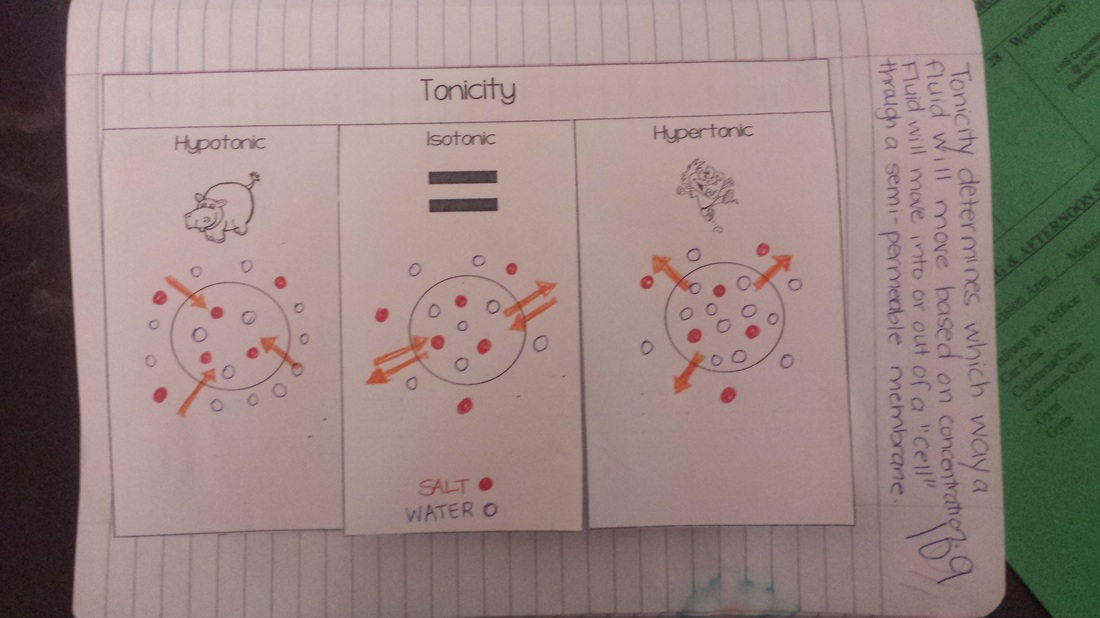
Today we reviewed osmosis and diffusion from last year before beginning to learn about tonicity. Students will need this knowledge to carry out our lab beginning tomorrow on how gummy bears react to different solutions. We watched a video about how tonicity can affect the human body, and that video and the notes are posted below. Students should also be preparing for our upcoming test on Wednesday that covers pages 79-103 in the science notebook!
Today we spent a good portion of our time going over problems from yesterday's classwork and homework. We have a quiz tomorrow on balancing equations. In addition, we also learned how to identify four different types of chemical equations. Check the notes below if you missed class!
Still confused on how to balance chemical equations? Here is a great tutorial that walks you through the process step by step!
Below are the answers to the classwork that we worked on today in class. Please check your answers to make sure you are CORRECT! Don't wait until the morning of our quiz to tell me you don't understand how to do it! I will post the answers to the individual work after I have checked your homework tomorrow! We have been working our way up to this standard FOREVER, but we have finally reached a point where we can start balancing equations. Many students got their first taste of this "fun" today. I can tell you we will have MUCH MORE practice early next week, but be prepared for a quiz on Wednesday. Any students who need extra help can either see me during 3rd period or meet with Mr. Jackson before school next week. Check out the notes below! ( I will post page 101 after all students have had a chance to try the challenge problem!)
Today we continued on with chemical reactions by learning about endothermic and exothermic reactions. We spent a little bit of time looking at the reactions in the classroom and talking about how everyday things like baking can be considered in one of these categories. One of the things we discussed was Elephant Toothpaste, an exothermic reaction. After we did our own less-concentrated classroom version, students watched the video below to see how a more-concentrated set of chemicals would change the reaction. This also gave us a chance to discuss different factors that lead to changes in reactions. Today we went back to review ionic and covalent bonding by participating in a class activity called "Bond with Classmates". Students were assigned either a positive or negative ion and were responsible for finding an alternate ion in the room to bond with. Students not only had to write the chemical formula for their bond, but they also had to work together to name their bonded atoms. (P.S.- I can't take credit for this great activity so you can find it here.)
|
AuthorI am an 8th grade Science teacher in Leeds, Alabama. This is my fourth year teaching science. As a teacher, I would love nothing more than to teach my kids to question the world around them and to never stop learning. Archives
May 2017
Categories
All
|






















 RSS Feed
RSS Feed