|
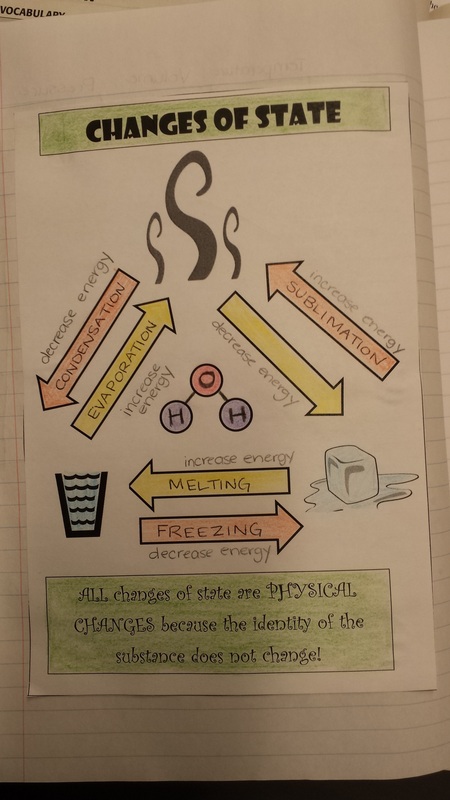
Delving into the physical property of state, today we discussed how to change between the different states: solid, liquid, and gas. We added a new graphic to our science notebook and explored the differences between exothermic states of change and endothermic states of change.
0 Comments
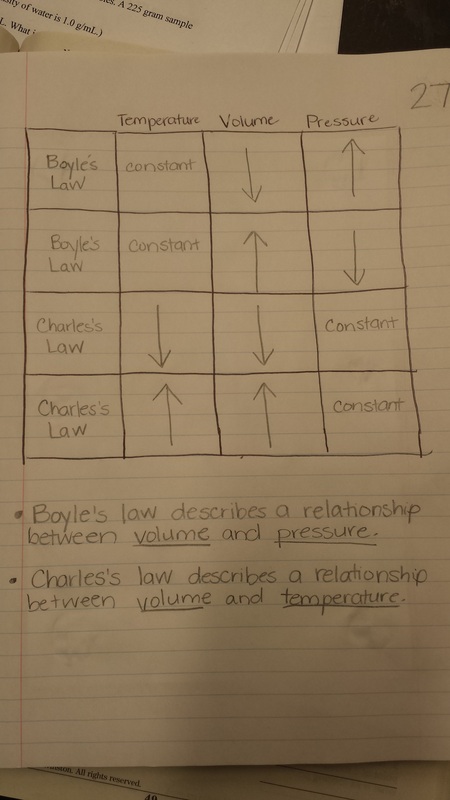
Today we discussed two gas behavior laws and how they impact the pressure, volume, and temperature of gases.
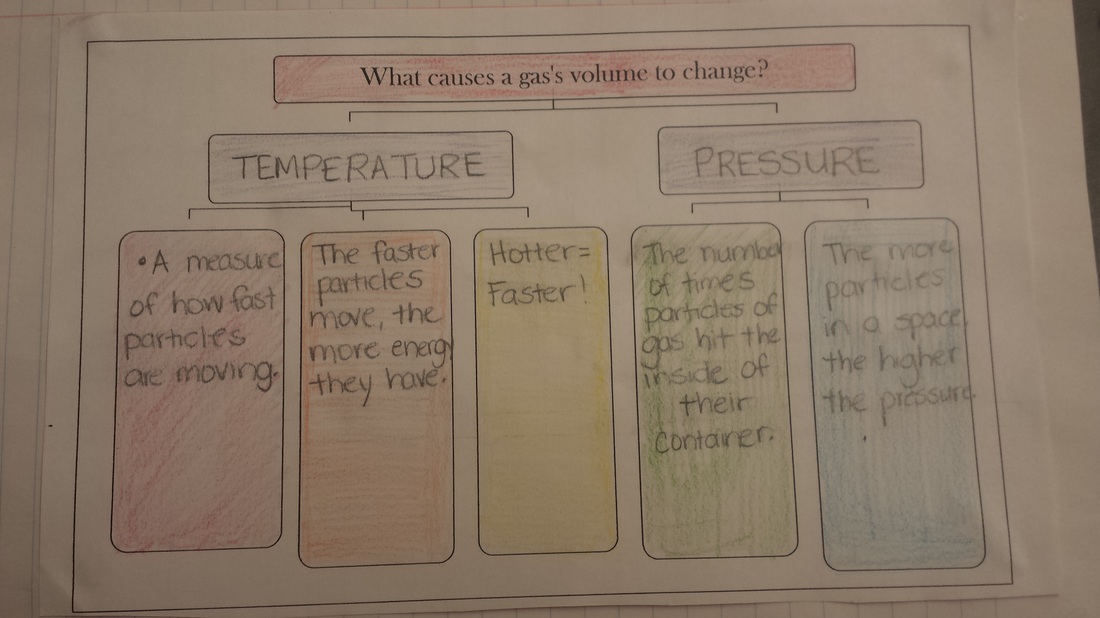
Today we started learning about the different states of matter and how they form based on particle interaction. We added the following graphic to our science notebook to help us remember the differences. We then started a gas behavior flowchart that showcases the ways in which a gas's volume can change.
We will have our first unit test on Wednesday, January 22! Please make sure that your notebook is updated as notebooks will also be checked on this day! Download a copy of the rubric if you missed it in class so you know what I am grading! I am also attaching a copy of the study guide in case you lost yours!
 Today in lab we explored properties of chemical changes. After discussing the differences between physical and chemical properties, we took a moment to learn about some of the signs of chemical changes by creating elephant toothpaste. This is a really cool experiment that can be recreated at home. Elephant Toothpaste Recipe
1. Put on the safety goggles and position the empty soda bottle in the middle of the foil pan in front of you.
2. Put the funnel in the top of the bottle and add the 1/2 cup of peroxide and 3-4 drops of food coloring 3. Add the squirt of dawn detergent to the bottle. 4. Pour the yeast mixture in the bottle and quickly remove the funnel! TA-DA! ELEPHANT TOOTHPASTE!  Density is a new concept for the students, and with that in mind, we have added a new graphic to our SN to help us learn how to calculate density. This "Density Triangle" will be helpful as students attempt to manipulate this formula on classwork and homework. Please view the video below if you need a refresher course on how to work the density triangle! In addition to the density triangle, please check the answers to the classwork below!
In addition to density, we also used these two class periods to cover chemical properties and changes. Once we covered chemical properties and chemical changes, we created a T-chart to compare different scenarios and determine whether the scenario depicted a physical change or a chemical change.
There was a quiz today! If you missed it, come see me 4th period!
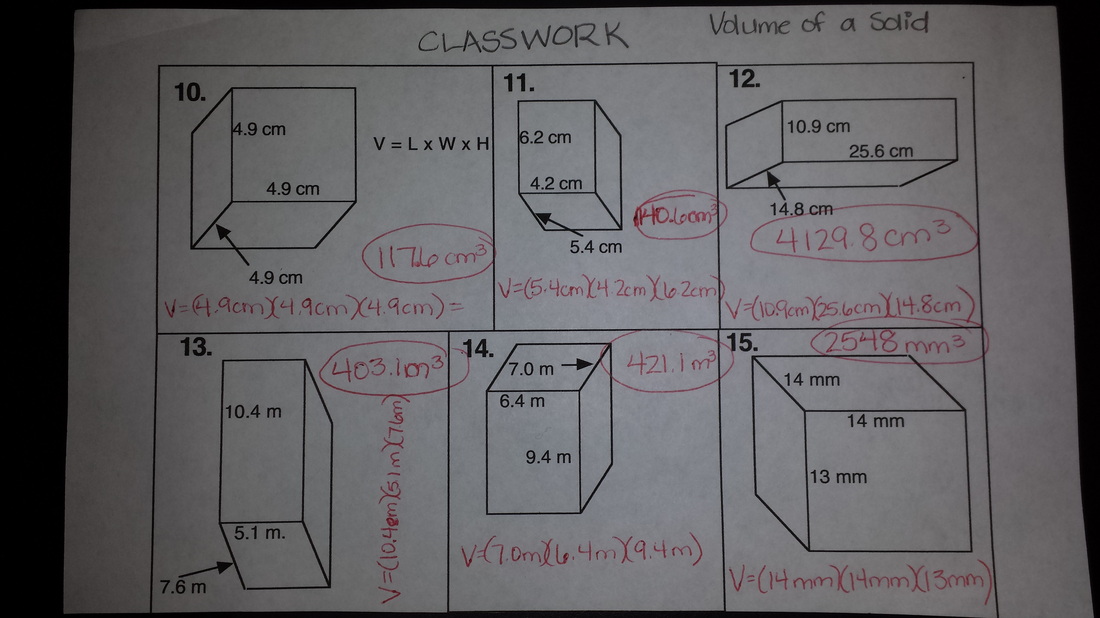
Today in class we finished up volume practice (see the answers to classwork below) and started exploring the physical properties of matter, such as solubility, state, and density. Students also explored the physical properties of matter last week as they tried to guess the identity of a mystery item by observing with their senses.
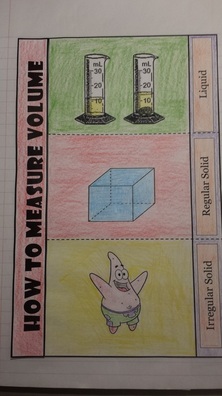
Today we practiced measuring volume on paper as it will be done on the upcoming test. Students have classwork for this glued into notebooks, and there is also a homework assignment on this for tonight! I am posting the answers to the classwork in case you need help with the homework and forgot your notebook! In addition to working with volume, we also reviewed mass and weight from last semester by creating a T-Chart of properties related to mass and weight. Here is the chart in case you missed it:
 Today we started learning about properties of matter with a quick mystery lab. Students were asked to make observations about an item in a sealed paper bag. Students could not open the bag, but they could touch, smell, shake, and listen to the object within. Students were then asked to draw a conclusion about the object's identity based on what they had observed and analyze how their observations led to that conclusion.
|
AuthorI am an 8th grade Science teacher in Leeds, Alabama. This is my fourth year teaching science. As a teacher, I would love nothing more than to teach my kids to question the world around them and to never stop learning. Archives
May 2017
Categories
All
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||















 RSS Feed
RSS Feed