|
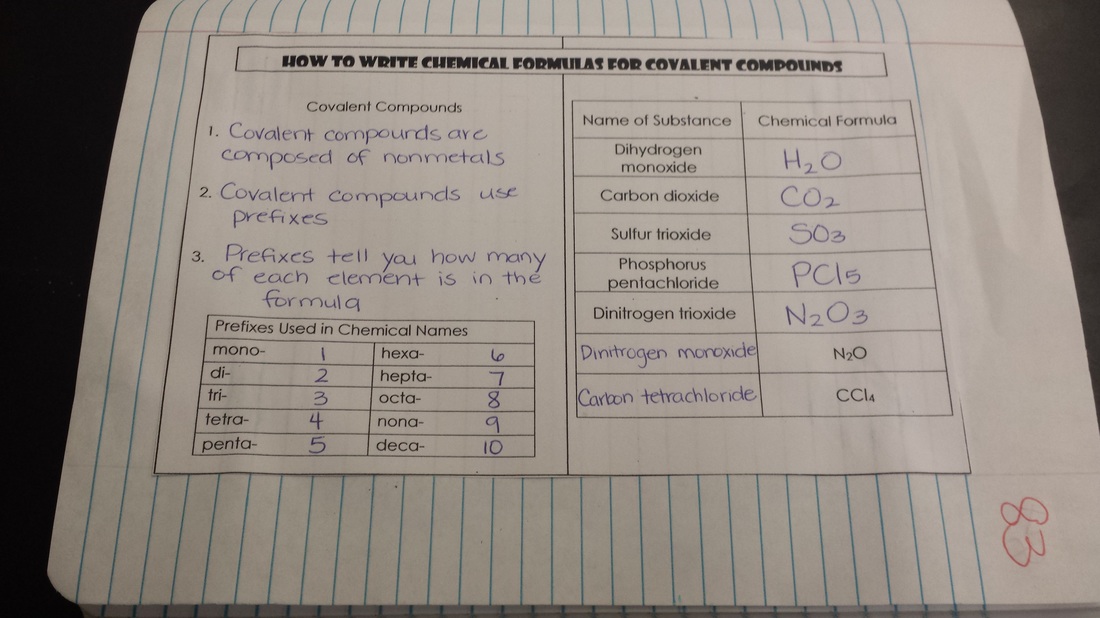
We are steadily working our way up to balancing chemical equations. Students have learned about chemical formulas and how to read them and name them as well as how to draw Lewis Structures to represent atoms. Check the notes below if you missed them!
0 Comments
We have been working in class on the different types of bonds and how to differentiate between them. We also went back to discover who was correct in the "Chemical Bonds" answers from page 73. There will be a quiz Wednesday on the different types of bonds as well as Electron Diagrams from last week!
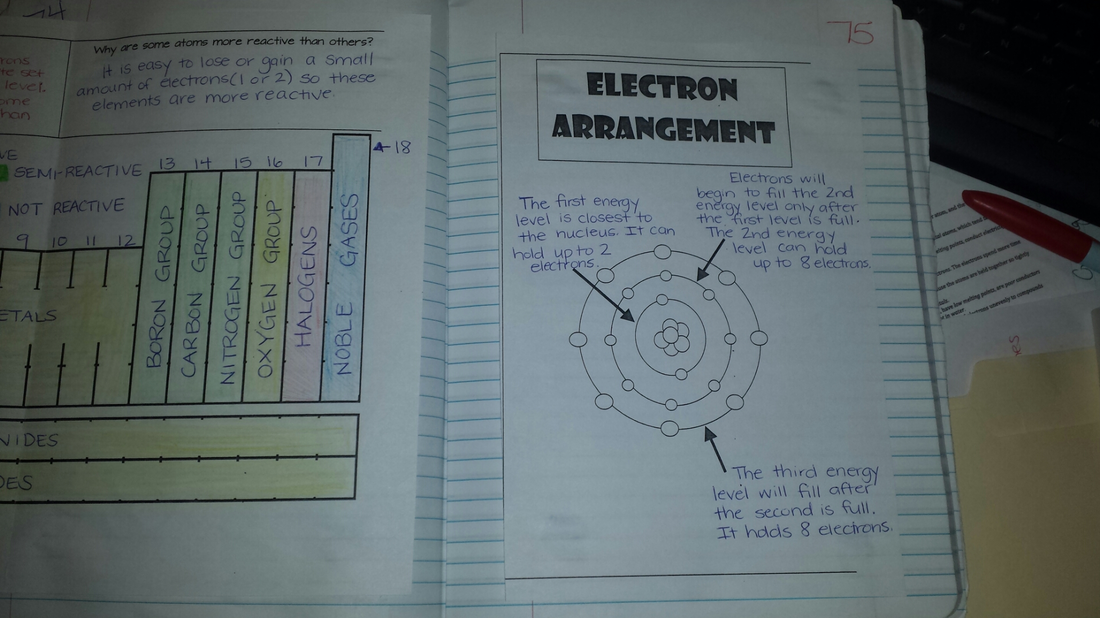
Students worked with diagramming electrons to help them understand where valence electrons and reactivity come from.
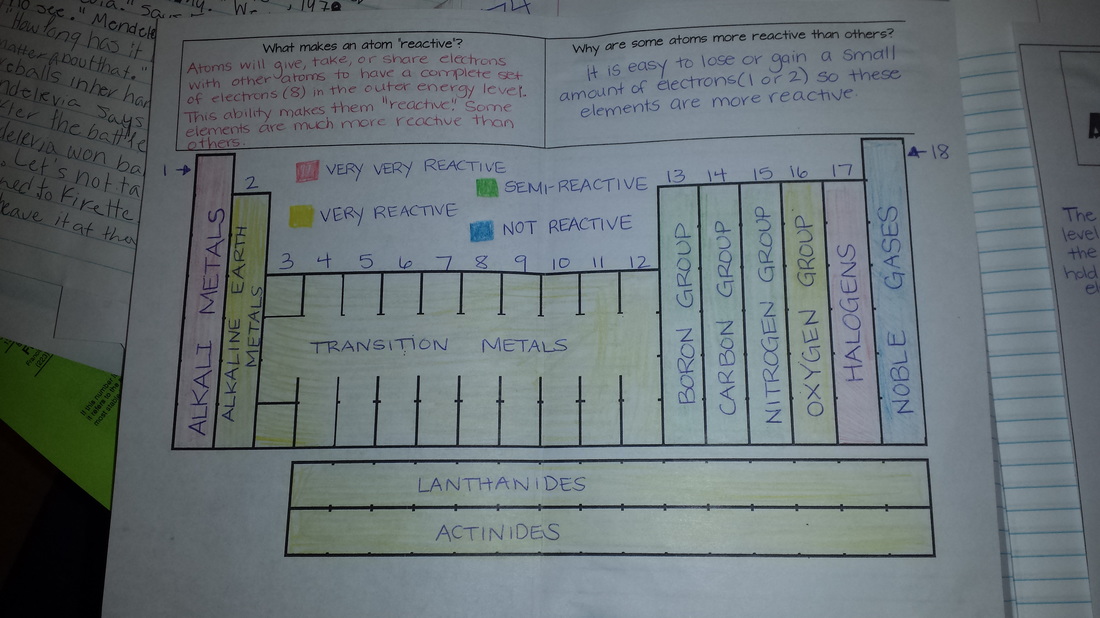
Today we colored the reactivity trends of the periodic table based on group. Students learned about how valence electrons impact the reactivity of specific elements. We then began to work on diagramming atoms and we will continue with this tomorrow.
Today we started Unit 3 (Chemical Bonding) by writing our essential questions and completing a "Chemical Bonds" probe. (We will come back and answer this later after we get more information!) We then did a few notes on the specifics of bonding. We will continue working on periodic table reactivity tomorrow!
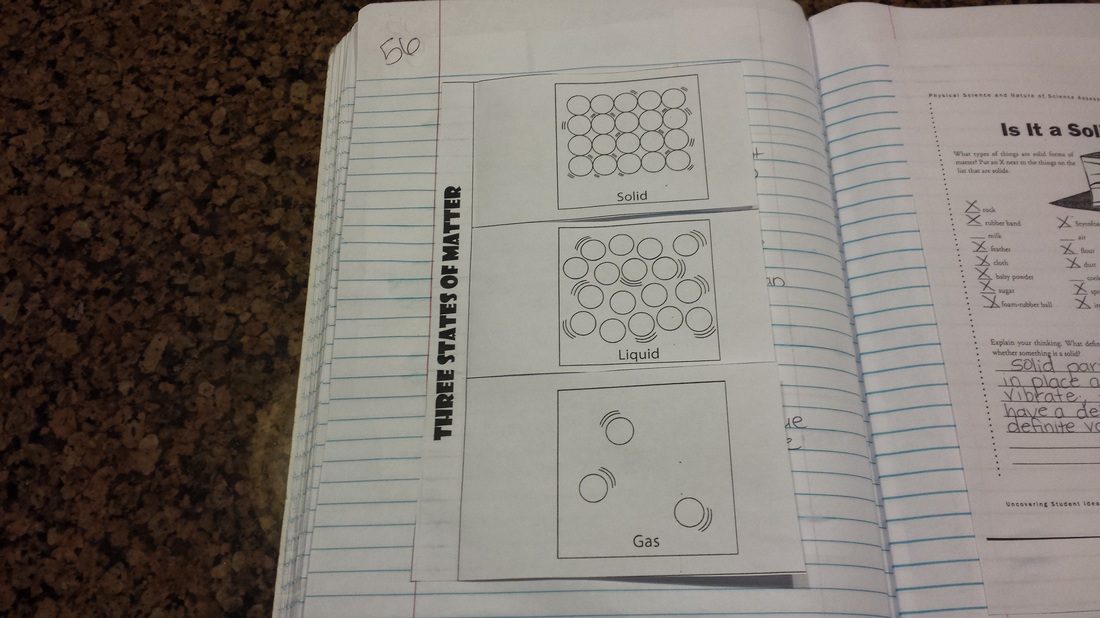
Today we worked with identifying physical and chemical properties and changes. Students also created a T-chart where they had to classify changes as chemical or physical. Our 1st 9 weeks exam will be Monday, October 19th. "Study guides" were glued into notebooks today. There will be a question and answer session regarding the study guide on Friday, so please be sure to look over it before then! Check out the notes below if you missed them in class! Today we finished discussing how the energy of particles is related to the state matter is in. Gases have the highest energy and solids have the lowest. Then we went back to discussing the bubble question from the other day. The correct answer to the question is.......LIQUID AND GAS! To make a bubble you have to blow (gas) into a liquid. So there is a liquid layer surrounding the gas. This led us into a great discussion about surface tension as we made GIANT bubbles in the classroom. I wish I had pictures but it is virtually impossible to snap a shot when you are the bubble maker! Check out the notes below if you missed them in class! Also check out the recipes for making giant bubbles at home! Something cool to do over fall break!
I have been having problems with some of my posts lately. Often they are not appearing at all on the site or parts are missing. If you happen to notice something that you need is missing, please email me and I will get it up for you! That being said, we had a great day in lab as we learned about sublimation. The kids were able to "play" with dry ice and examine what happens to it under different conditions. Please check the notes below if you missed them. Yesterday we worked on the differences between solids, liquids, and gases. Students were then asked if they thought bubbles were a solid, liquid, or gas. More on that tomorrow! Today we started research for the periodic table project. Project details are listed below. Most of the research and development for this project will be completed in class, but students who do not use their time wisely or miss class will need to complete it at home. This project is due on October 16th! I will give 5 BONUS points to anyone who turns it in on Thursday, October 15th! 1. Identify the number of protons in Phosphorus.
The atomic number of Phosphorus is 15 so that tells me there are 15 protons in the nucleus. 2. How many electrons are in Potassium? Potassium's atomic number is 19 which is equal to the number of electrons. 3. How many neutrons are in an Argon atom? Argon's mass is 40 (39.95) and it's atomic number is 18 so when I subtract, I get 22 neutrons. 4. What is the atomic mass of Aluminum? According to the periodic table, the mass is 26.98. 5. What is the atomic number of Sodium? According to the periodic table, the atomic number is 11. 6. What is the chemical symbol of Mercury? Hg 7. What is the name for Xe? Xenon 8. Which element has the greatest mass, Fluorine or Sulfur? Fluorine's mass is 19 while Sulfur's is 32.07 so Sulfur has the greatest mass. 9. Which element has the most protons, Silicon or Beryllium? Silicon's atomic number is 14 and Beryllium's is 4 so Silicon has the most protons. 10. Which element has the most neutrons, Helium or Carbon? Using Mass-atomic number I found that Helium has 2 neutrons while Carbon has 6. **Also make sure you study the location of the metalloids, metals, and nonmetals! ** |
AuthorI am an 8th grade Science teacher in Leeds, Alabama. This is my fourth year teaching science. As a teacher, I would love nothing more than to teach my kids to question the world around them and to never stop learning. Archives
May 2017
Categories
All
|































 RSS Feed
RSS Feed