|
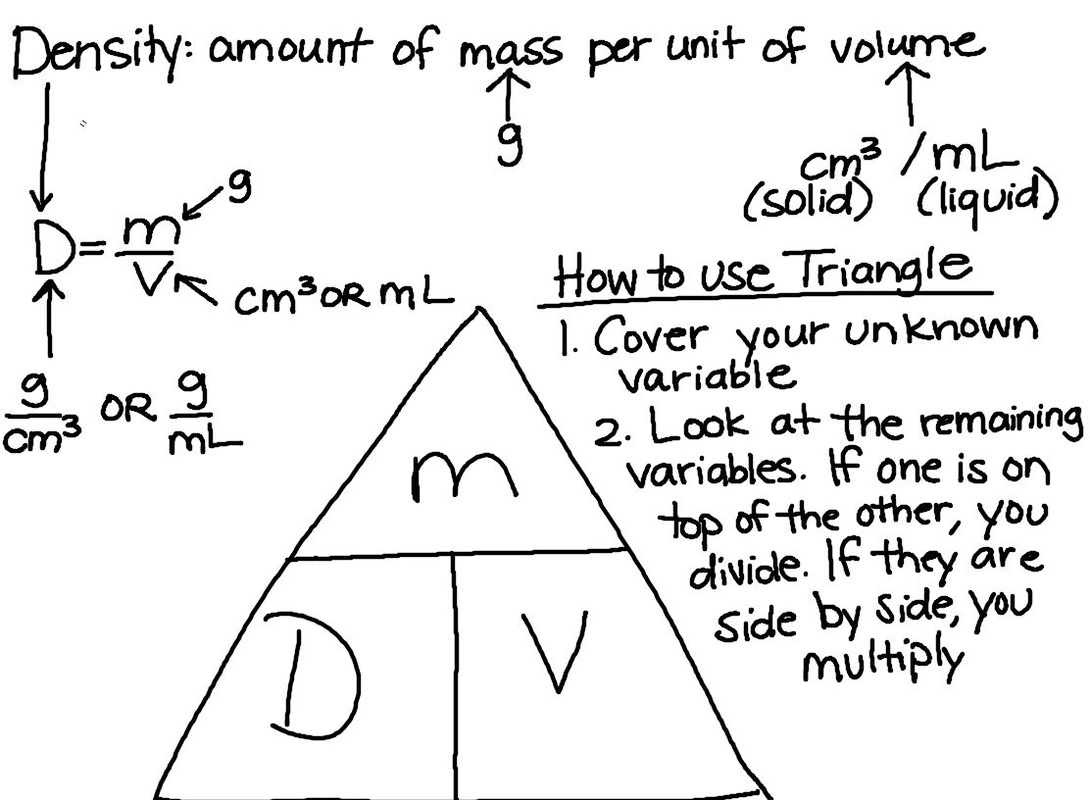
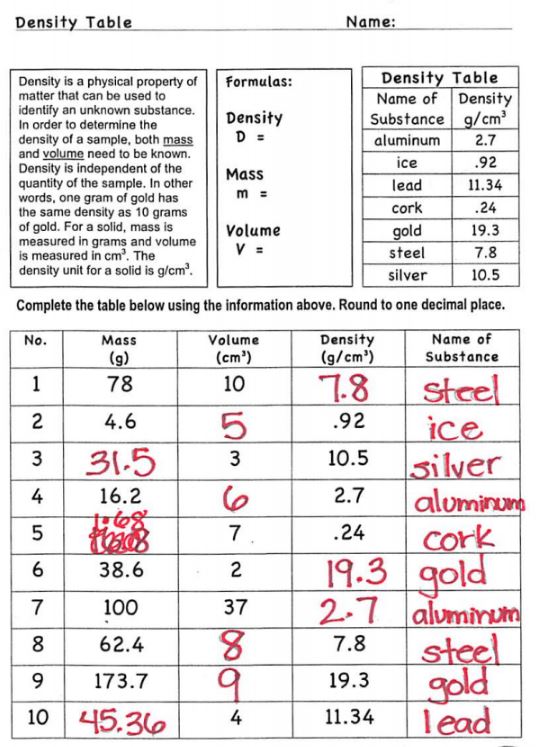
Today we worked on solving the density equation for any variable in the equation using the triangle. We will continue working on this Tuesday before we review with the test on Wednesday!
0 Comments
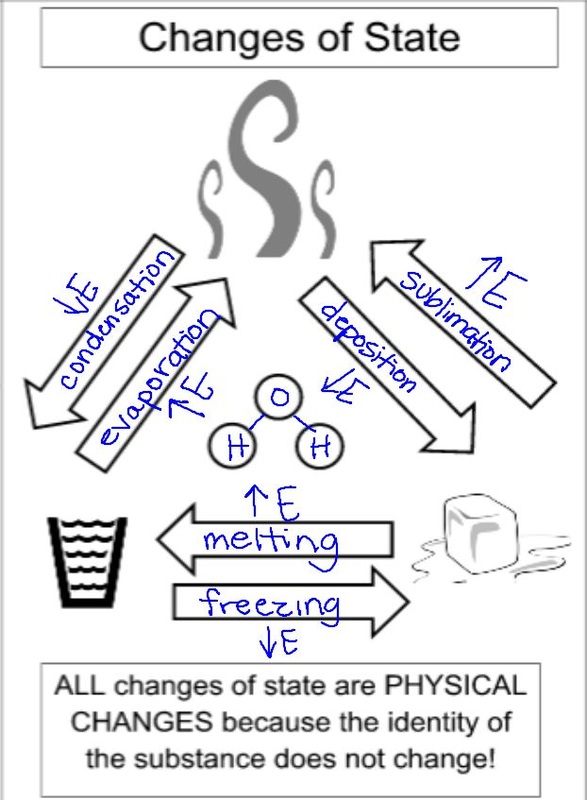
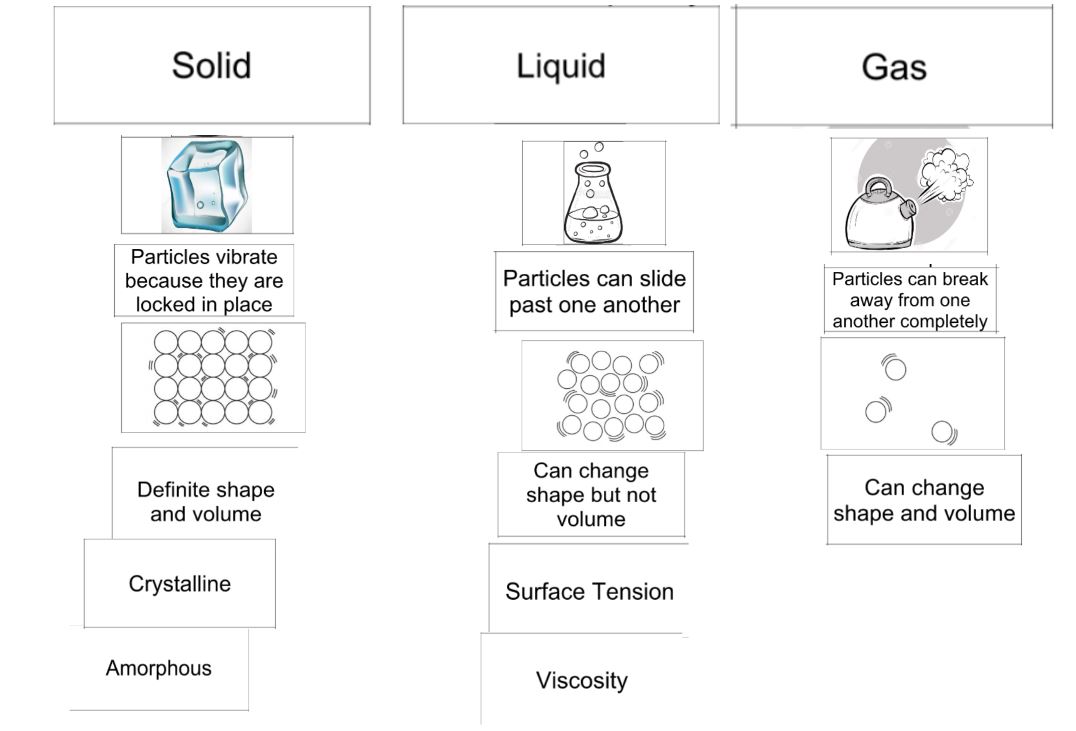
I apologize for the lack of posting yesterday, but I completely forgot! Yesterday we did a lab with dry ice where students made observations and learned that changing states of matter is a physical change. We also completed a graphic to help them understand the names of the phase changes and the relationship of the energy in the particles. Today we worked with the differences in physical and chemical properties and changes. I am posting the worksheet (with answers) from class below for students who were absent.
Today students did a little informational reading to help them determine the differences between physical and chemical properties and changes. They completed a worksheet in class with their group using an outline and the textbook, as well as their ideas as a group. They were then given a creative writing assignment to begin. This creative writing assignment is due on WEDNESDAY at the beginning of class and is posted below for students who missed it!
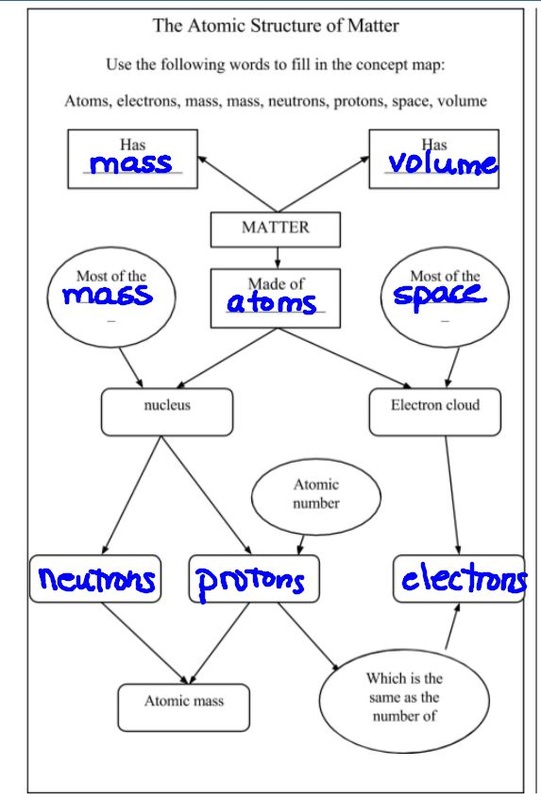
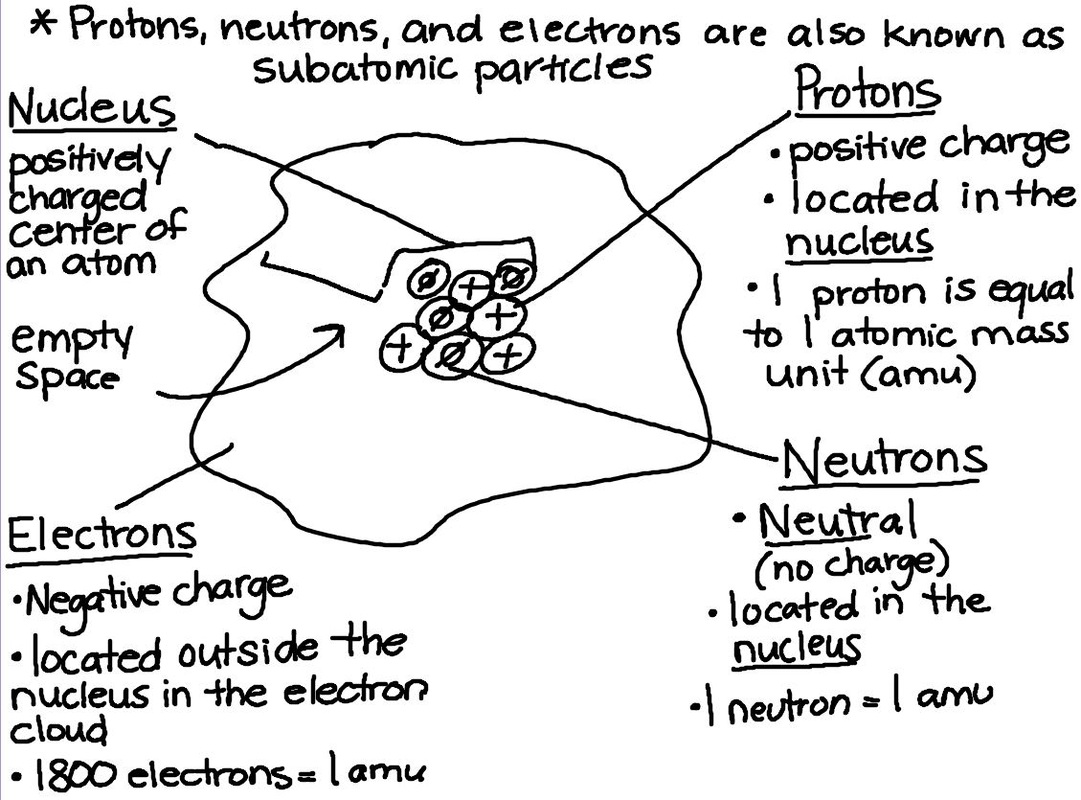
Today we took some time to review atomic structure before starting states of matter. We are going to continue with states of matter on Monday before we move on to physical and chemical properties and changes.
Today students took a pop quiz on atomic structure and finding the number of protons, neutrons, and electrons using the periodic table. Ms. Hendricks will be in here tomorrow going over Cornell notes and study habits, so we will continue with Bohr Models on Friday!
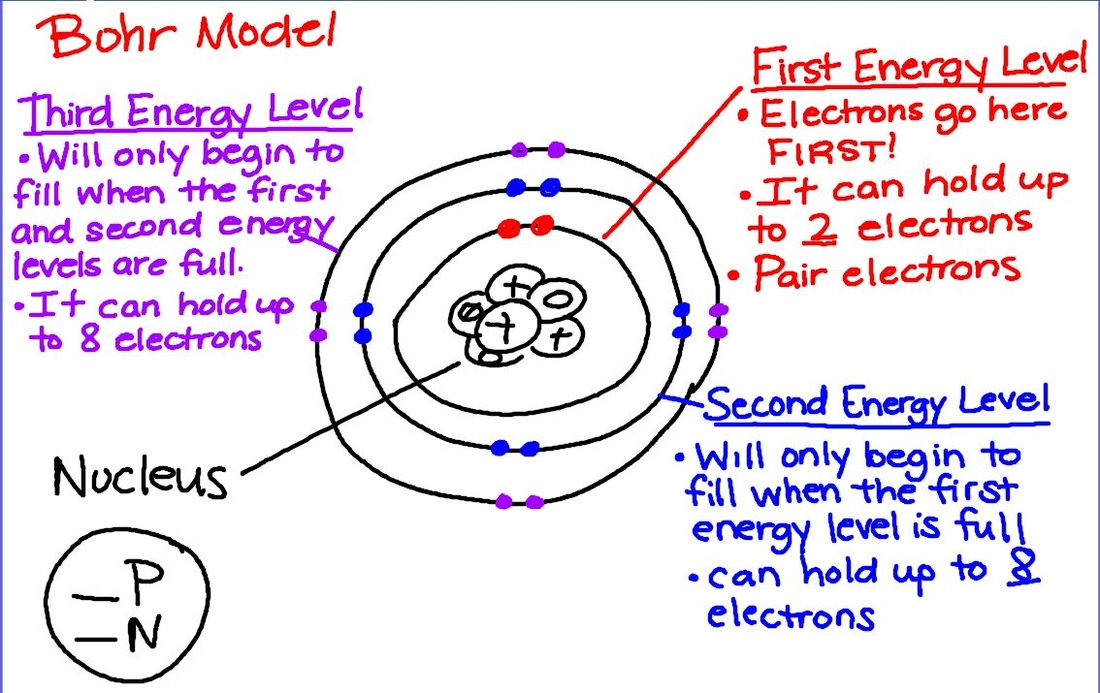
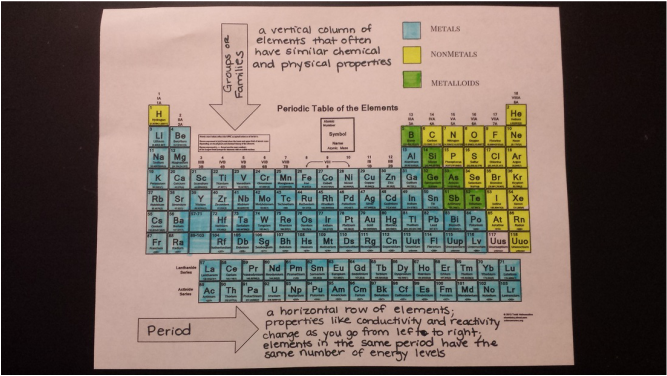
Today we began mapping atomic structure with Bohr models. We modeled this concept with beans, and tomorrow we will begin to draw them on paper. Don't forget that Atoms Family Homework is due tomorrow! Today we color coded the periodic table to represent metals, nonmetals, and metalloids. Students learned that the table is composed of mostly metals and that nonmetals are mostly gases at room temperature. We will continue with Bohr Models tomorrow!
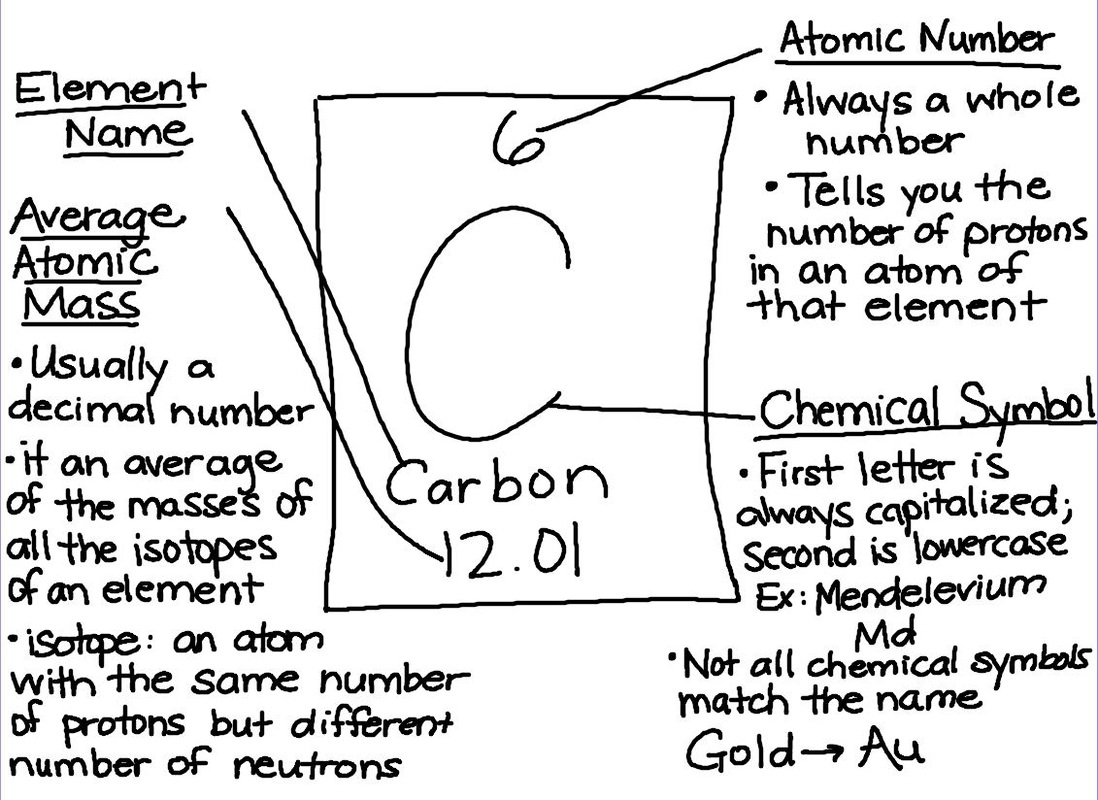
Today we drew diagrams of an atom and one of the elements on the periodic table. From there, students were presented with their first PERIODIC TABLE OF ELEMENTS! We just started using them today, but they learned how to identify the number of protons, neutrons, and electrons using the table.
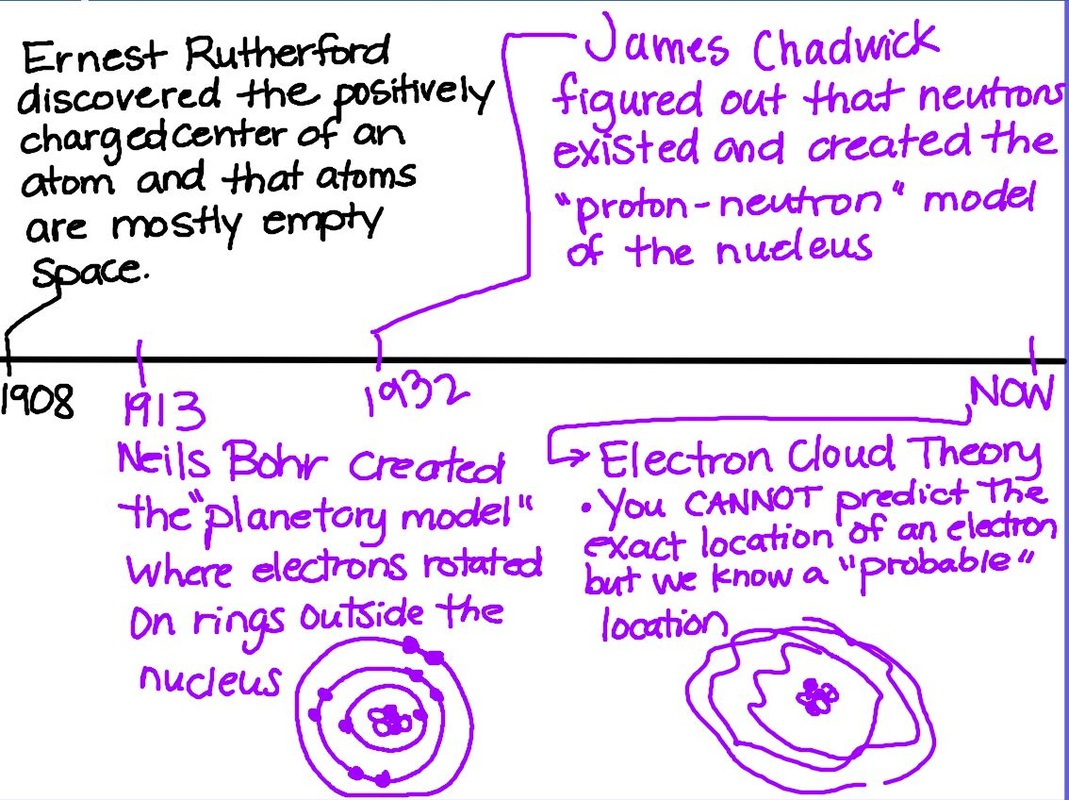
https://phet.colorado.edu/sims/html/rutherford-scattering/latest/rutherford-scattering_en.html Today we started off with a simulation comparing Thomson's and Rutherford's working, allowing students to "see" why Rutherford got the results he did in his experiments. We then finished the atomic theory timeline (pop quiz sometime this week!) and moved on to notes about elements and the periodic table! Second Half of the Timeline First part of elements and periodic table notes
Today in science we work on identifying shapes under a piece of cardboard using only a marble. Similar to Rutherford's work, students had to deduce the shape based on how the marble bounced off of it. Students made observations and educated guesses about the shape before we revealed them at the end. We will continue with a simulation on Rutherford's experiment tomorrow!
|
AuthorI am an 8th grade Science teacher in Leeds, Alabama. This is my fourth year teaching science. As a teacher, I would love nothing more than to teach my kids to question the world around them and to never stop learning. Archives
May 2017
Categories
All
|
||||||||||||||||||









 RSS Feed
RSS Feed